This web page was produced as an assignment for Genetics 564, an undergraduate course at UW-Madison.
Conclusions
Introduction
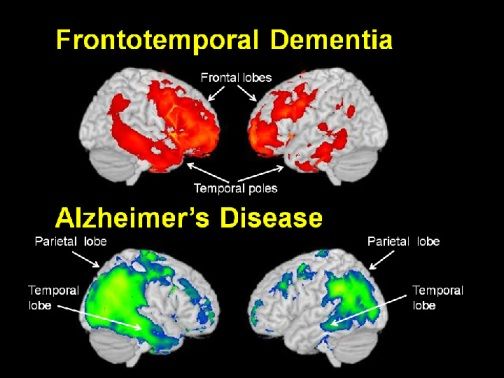
Fronto-temporal Dementia is a neurodegenerative disease that results in early onset symptoms of behavior, language, and learning difficulties. Also included in the FTD phenotype is a decrease in Grey Matter (GM) Density, or the number of active cells in the brain. There is no known cure or treatment for FTD, so further research is required to learn more information about the disease. MAPT encodes for the Tau protein and is associated with the FTD phenotype when mutated. There are several mutations possible, most in and around exon 10. After initial research, it was determined that there are four important regions of the MAPT protein sequence (called protein domains) that correspond to the exon 10 area. Because of the proximity of the protein domains to the genomic exons, it is hypothesized that mutations in the MAPT gene sequence could alter the post-transcriptional splicing of the gene, and therefore alter the Tau protein levels or function.
Further research showed the MAPT gene encodes for Tau proteins that are known to stabilize microtubules for cell differentiation and dividing. It is hypothesized that when the MAPT gene is mutated, it alters the levels of Tau protein made. With increased levels of Tau protein, microtubules in the cell become less stable, leading to decreased cell differentiation. This will have a negative effect on brain development and repair, leading to decreased brain cell density and therefore a decrease in the GM density. Given this hypothesis, the brain deterioration seen in the FTD phenotype is thought to be caused by the cascading effects of a mutation in the MAPT gene.
In order to gain more information about the FTD phenotype and test these hypotheses, various biotechnological techniques will be used. First, the levels of Tau protein will be compared between wild-type (normal) mice and mice with a mutation in the MAPT gene. Second, chemical genetic analysis will be used to find molecules that can bind to and reduce the amount of Tau proteins in the brain. Last, the learning and behavior of mice with a MAPT mutation will be compared to that of normal mice using a psychological test.
Further research showed the MAPT gene encodes for Tau proteins that are known to stabilize microtubules for cell differentiation and dividing. It is hypothesized that when the MAPT gene is mutated, it alters the levels of Tau protein made. With increased levels of Tau protein, microtubules in the cell become less stable, leading to decreased cell differentiation. This will have a negative effect on brain development and repair, leading to decreased brain cell density and therefore a decrease in the GM density. Given this hypothesis, the brain deterioration seen in the FTD phenotype is thought to be caused by the cascading effects of a mutation in the MAPT gene.
In order to gain more information about the FTD phenotype and test these hypotheses, various biotechnological techniques will be used. First, the levels of Tau protein will be compared between wild-type (normal) mice and mice with a mutation in the MAPT gene. Second, chemical genetic analysis will be used to find molecules that can bind to and reduce the amount of Tau proteins in the brain. Last, the learning and behavior of mice with a MAPT mutation will be compared to that of normal mice using a psychological test.
What should be studied?
|
Aim 2: Determine chemical genetic compounds that can alter Tau protein levels
Chemical genetic analysis of the Tau protein has yet to be completed in regards to Fronto-temporal dementia. This type of analysis can use two different types of methods to determine possible small molecules that can bind to a protein in question. After finding small molecules that may bind to the protein, a protein-binding assay is then completed to determine exactly which small molecules bind directly to the protein. For the Tau protein, first a diversity-oriented library should be used in order to obtain the most possible small molecules for the analysis. After, a protein-binding assay will be completed to find the small molecules that bind directly to Tau. After, analysis should be completed to determine which of these molecules could decrease the levels of Tau in the brain cells. By decreasing the Tau proteins, it could decrease the number of Tau protein tangles, therefore keeping microtubules stabilized and allowing correct cell differentiation.\ Hypothesis: The small molecules found to bind to Tau proteins may be used for targeted drug therapy because the chemical binding will prevent the Tau proteins from aggregating. This will, in turn, stabilize microtubules and lead to normal cell differentiation. |
|
Aim 3: Evaluate changes in learning and behavior based on an induce mutation in the Tau protein
In order to determine if the phenotypic changes seen in FTD are caused by mutations in the MAPT gene, the CRISPR-Cas9 system will be used to induce mutations in either exon 10 or intron 10. From there, the learning and behavior can be tested with a psychology test called the Hebb-Williams maze. Hypothesis: If mutations in the MAPT gene are the cause of FTD, the mice should fail to complete the exercise well and fail the test. The wild-type mice (without a mutation in exon 10 or intron 10) should pass the exam as normal. |
What are some future directions for FTD research?
|
After researching FTD for several months, it is clear that more laboratory research should be completed in the near future. Although several things are known about the disease, there are far too many unknown components of the disease to have acceptable diagnosis and treatment.
In terms of further research, there are several aspects that should be looked at closely. New neuroimaging techniques are being developed that look specifically at Tau protein levels. If this is the case, it can be useful to use this type of machine to measure protein levels and diagnose FTD at an earlier time. More research into the causes and phenotypes of FTD can give more insight into the differences between FTD and Alzheimer's Disease. This information can be imperative to a correct diagnosis and further care and treatment. Further research, especially the chemical genetics analysis, will give researchers more insight into possible treatments for FTD. Using small molecule techniques, it is possible the levels of Tau protein can be decreased and therefore alter the phenotype of FTD patients. Figure 10. Examples of pharmaceuticals that could be created based on chemical genetic analysis.
|
Final Course Presentation (updated 5/5/15)
| fordfinaltalk4-30-15.pdf | |
| File Size: | 8674 kb |
| File Type: | |
Site built by Kassandra Ford
Genetics 564, Spring 2015
University of Wisconsin-Madison
Site last updated: 5/13/2015
Genetics 564, Spring 2015
University of Wisconsin-Madison
Site last updated: 5/13/2015