This web page was produced as an assignment for Genetics 564, an undergraduate course at UW-Madison.
What is Fronotemporal Dementia?
Frontotemporal Dementia (FTD) is a degenerative disease associated with shrinking of the frontal lobes of the brain. It has several names: Frontotemporal Dementia, Frontotemporal Lobar Degeneration, and Pick's Disease. There are generally two categories of the disease. The first is associated with changes in social behavior, the second is associated with language deficiencies. They are called, respectively, behavioral-variant frontotemporal dementia and primary progressive aphasia [1]. Many patients with the disease maintain physical capabilities and some memory. It is equally likely in men and women, and is most commonly diagnosed between 50 and 60 years of age.[2] The prevalence of FTD is approximately 15 in 100,000 people aged 45 and 60. Although there is no known cure for the disease at this time, research is being done by multiple organizations to look into the genetic components of the disease.[3] The disease itself is not fatal, but predisposes patients to illnesses like pneumonia, infection, or injury. [2]
Frontotemporal Dementia (FTD) is a degenerative disease associated with shrinking of the frontal lobes of the brain. It has several names: Frontotemporal Dementia, Frontotemporal Lobar Degeneration, and Pick's Disease. There are generally two categories of the disease. The first is associated with changes in social behavior, the second is associated with language deficiencies. They are called, respectively, behavioral-variant frontotemporal dementia and primary progressive aphasia [1]. Many patients with the disease maintain physical capabilities and some memory. It is equally likely in men and women, and is most commonly diagnosed between 50 and 60 years of age.[2] The prevalence of FTD is approximately 15 in 100,000 people aged 45 and 60. Although there is no known cure for the disease at this time, research is being done by multiple organizations to look into the genetic components of the disease.[3] The disease itself is not fatal, but predisposes patients to illnesses like pneumonia, infection, or injury. [2]
Overview of Frontotemporal Dementia |
The impacts of FTD on behavior and language |
|
YouTube video with the general overview of FTD. From the University of California, San Fransisco. Video Credit
|
YouTube video with the impacts of FTD on behavior and language. From the University of California, San Fransisco. Video Credit
|
What is the cause of FTD?
Previous research has connected FTD to mutations in multiple genes, one of the most common genes being the Microtubule-associated Protein Tau (MAPT) gene. The MAPT gene encodes for the Tau protein in the brain. Mutations in the MAPT gene can lead to over-expression of the Tau proteins. These mutations and phenotype have been found in patients with FTD.
Previous research has connected FTD to mutations in multiple genes, one of the most common genes being the Microtubule-associated Protein Tau (MAPT) gene. The MAPT gene encodes for the Tau protein in the brain. Mutations in the MAPT gene can lead to over-expression of the Tau proteins. These mutations and phenotype have been found in patients with FTD.
The gene basics
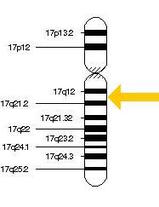
The MAPT gene is associated with the formation of the tau proteins in the brain. It is located on Chromosome 17 and has 15 exons [4]. There is alternative splicing, which makes six different isoforms of the tau protein in the human brain. [5] In young, developing humans, the 3R isoform dominates, while adult brains have an equal combination of 3R and 4R isoforms. This ratio is thought to be imperative to maintaining neural function and preventing dementia during adult life. [5]
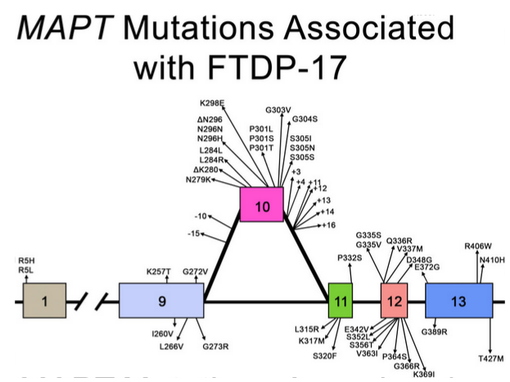
Mutations: Mutations have been found in exons 1, and 9-13, as well as an intron (10). The alternative mutations and exons associated with those mutations are shown below in Figure 3. These mutations have a common result: affecting the alternative splicing of exon 10, creating an excess of 4R tau. Exon 10 normally creates a stem-loop, but this is altered with any of these mutations. Some mutations are more detrimental than others, and are associated with immense over-expression of the Tau proteins. [5]
The MAPT gene is associated with the formation of the tau proteins in the brain. It is located on Chromosome 17 and has 15 exons [4]. There is alternative splicing, which makes six different isoforms of the tau protein in the human brain. [5] In young, developing humans, the 3R isoform dominates, while adult brains have an equal combination of 3R and 4R isoforms. This ratio is thought to be imperative to maintaining neural function and preventing dementia during adult life. [5]
Mutations: Mutations have been found in exons 1, and 9-13, as well as an intron (10). The alternative mutations and exons associated with those mutations are shown below in Figure 3. These mutations have a common result: affecting the alternative splicing of exon 10, creating an excess of 4R tau. Exon 10 normally creates a stem-loop, but this is altered with any of these mutations. Some mutations are more detrimental than others, and are associated with immense over-expression of the Tau proteins. [5]
Figure 1. Human chromosome 17, with the location of the MAPT gene highlighted with an arrow. Photo Credit
|
The protein basics
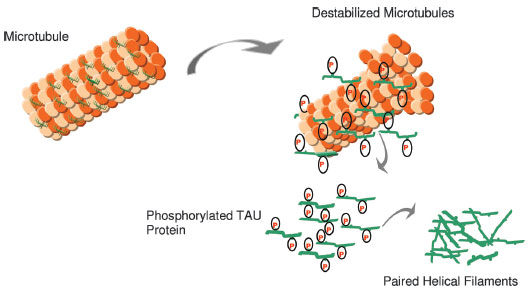
Normal Function: The Tau proteins bind to and stabilize microtubules in eukaryotic cells. Microtubules are essential for processes like cell differentiation, mitosis, and meiosis [4]. They are comprised of tubulin and form long chains. Based on this, the Tau proteins are associated with correct cell differentiation and proliferation in eukaryotic cells. Abnormalities: When the Tau proteins are over-expressed, the movement around the cells by microtubules is affected. In particular, an excess of Tau protein leads to congregations of the proteins into a complex called a neurofibrillary tangle. Because the Tau proteins are aggregating in these tangles, they are no longer stabilizing the microtubules in the cell. In essence, when there is too much Tau protein in eukaryotic cells, microtubules are unable to function normally and cell differentiation is negatively affected. |
Figure 2 (above). The structure of microtubules. Normally stabilized by the TAU protein, but when the TAU proteins are phosphorylated, the microtubules are destabilized. Photo credit
Figure 3 (below). The known mutations to the MAPT gene. Many are found in exon 10, but there are other mutations possible. Photo credit |
Diagnosis and Management
There are various tests used to determine if a patient has Frontotemporal Dementia, or should be diagnosed with a different neurological disorder. These tests include blood work, neurological exams, Electroencephalograms (EEG), Electromyography (EMG), lumbar puncture, CT or CAT scan, MRI, PET scans, Functional MRI, or a SPECT scan. Some of these may be familiar to the general public, while others are more specialized or non-invasive. More information about each test can be found by clicking on the test of interest. A combination of any or all of these tests may be used, depending on the severity of the case. In many instances, a neuropsychological test is also administered. This evaluates memory, concentration, and language and can be used to differentiate between disorders like FTD and Alzheimer's disease [2].
As stated previously, at this time there is no known treatment for Fronototemporal dementia. Symptoms can be managed and supportive care is imperative. This disorder not only affects the person living with the condition, but the friends and family surrounding that individual.
As stated previously, at this time there is no known treatment for Fronototemporal dementia. Symptoms can be managed and supportive care is imperative. This disorder not only affects the person living with the condition, but the friends and family surrounding that individual.
REFERENCES
Cover Photo Credit
[1] Riedl, L., Mackenzie, I., Förstl, H., Kurz, A., & Diehl-Schmid, J. (n.d.). Frontotemporal lobar degeneration: Current perspectives. Retrieved February 1, 2015, from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3928059/
[2] The Association for Frontotemporal Degeneration
[3] National Institute of Neurological Disorders and Stroke
[4] Rademakers, R., Cruts, M., & Van Broeckhoven, C. (2004, May 7). The Role of Tau (MAPT) in Frontotemporal Dementia and Related Tauopathies. Retrieved February 15, 2015, from http://onlinelibrary.wiley.com/store/10.1002/humu.20086/asset/20086_ftp.pdf?v=1&t=i640wdvb&s=883c5ef03f70d555bd98baaa7fa1368f93d6069b
[5] Ghetti, B., Oblak, A., Johnson, K., Dickerson, B., & Goedert, M. (2015, February 1). Invited review: Frontotemporal dementia caused by microtubule-associated protein tau gene (MAPT) mutations: A chameleon for neuropathology and neuroimaging. Retrieved February 18, 2015, from http://onlinelibrary.wiley.com/doi/10.1111/nan.12213/epdf
Cover Photo Credit
[1] Riedl, L., Mackenzie, I., Förstl, H., Kurz, A., & Diehl-Schmid, J. (n.d.). Frontotemporal lobar degeneration: Current perspectives. Retrieved February 1, 2015, from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3928059/
[2] The Association for Frontotemporal Degeneration
[3] National Institute of Neurological Disorders and Stroke
[4] Rademakers, R., Cruts, M., & Van Broeckhoven, C. (2004, May 7). The Role of Tau (MAPT) in Frontotemporal Dementia and Related Tauopathies. Retrieved February 15, 2015, from http://onlinelibrary.wiley.com/store/10.1002/humu.20086/asset/20086_ftp.pdf?v=1&t=i640wdvb&s=883c5ef03f70d555bd98baaa7fa1368f93d6069b
[5] Ghetti, B., Oblak, A., Johnson, K., Dickerson, B., & Goedert, M. (2015, February 1). Invited review: Frontotemporal dementia caused by microtubule-associated protein tau gene (MAPT) mutations: A chameleon for neuropathology and neuroimaging. Retrieved February 18, 2015, from http://onlinelibrary.wiley.com/doi/10.1111/nan.12213/epdf
Site built by Kassandra Ford
Genetics 564, Spring 2015
University of Wisconsin-Madison
Site last updated: 5/13/2015
Genetics 564, Spring 2015
University of Wisconsin-Madison
Site last updated: 5/13/2015